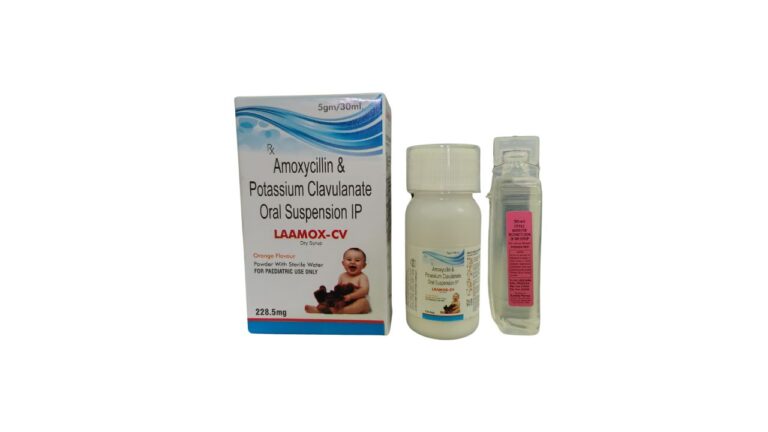
What is Alu Alu packing? | Best ALU ALU packing machine in India

Alu Alu packing (cold-form foil blister) is a high‑barrier blister format where both forming web and lidding are aluminium‑based films, typically OPA/Al/PVC laminate plus aluminium lidding foil. It provides near‑absolute protection against moisture, oxygen and light, making it ideal for…


![50-Point Pharma Plant Acquisition Checklist: [2026 Updated] GMP Due Diligence Guide" Pharma Plant Acquisition Checklist for GMP Due Diligence](https://laafon.com/wp-content/uploads/2026/04/Pharma-Plant-Acquisition-Checklist-768x403.jpg)