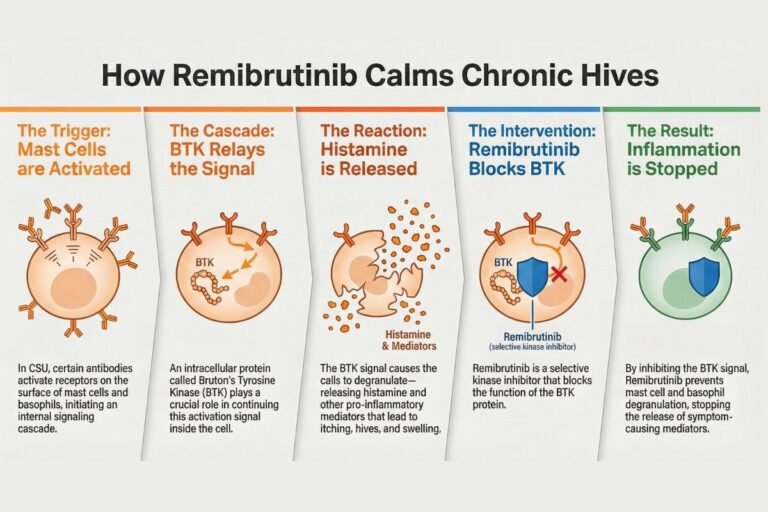
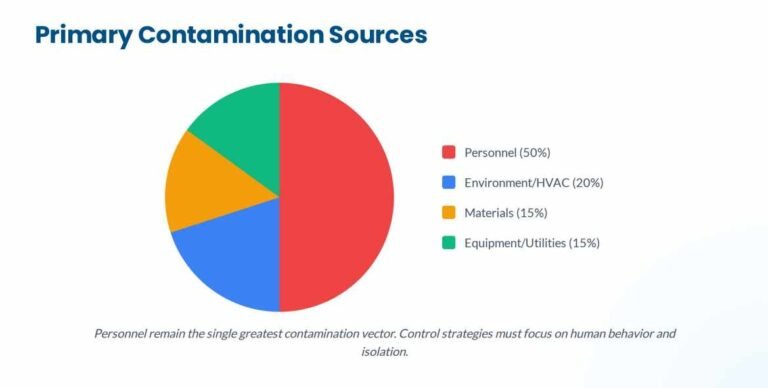
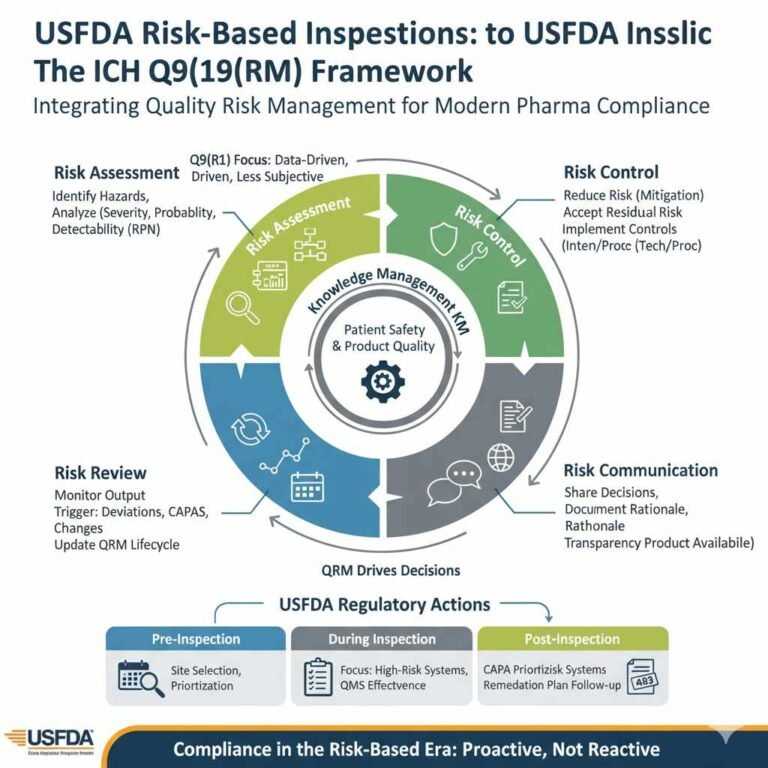
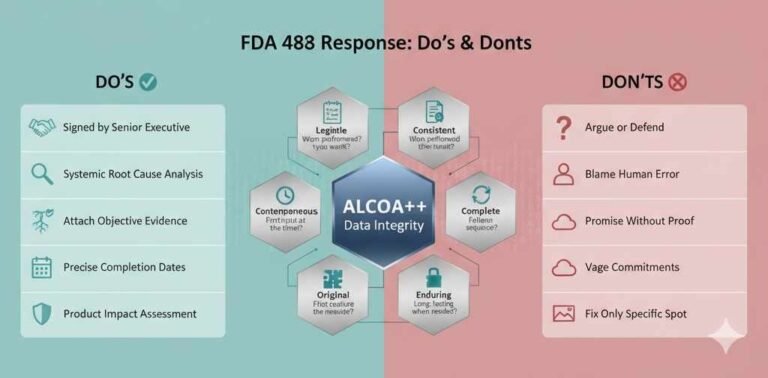
Voyxact (sibeprenlimab-szsi) FDA Approval: Efficacy, Dosage & Safety for IgAN

On November 25, 2025, the U.S. Food and Drug Administration (FDA) granted accelerated approval to VOYXACT (sibeprenlimab-szsi), a novel biologic therapy for adults with primary immunoglobulin A nephropathy (IgAN)[3]. Developed by Otsuka Pharmaceutical Company, Ltd., this approval marks a significant…