Folflox-OZ Tablet (Ofloxacin 200 mg + Ornidazole 500 mg): Uses, Dose and Safety for Professionals
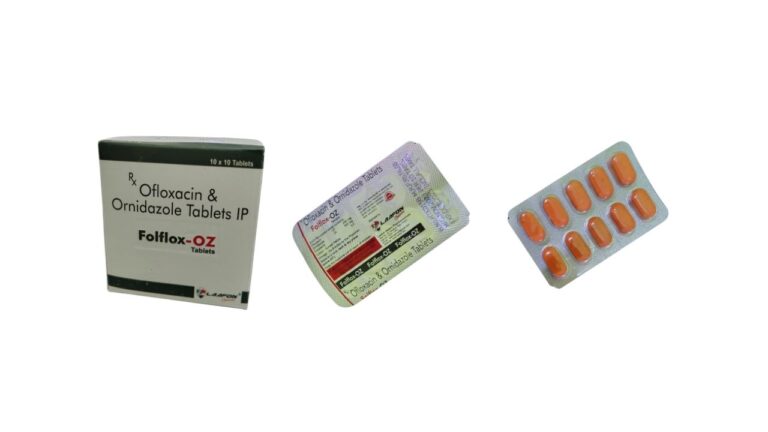
Folflox‑OZ is a fixed‑dose combination tablet containing ofloxacin 200 mg and ornidazole 500 mg, used mainly in adults for diarrhoea due to mixed bacterial–protozoal or aerobic–anaerobic infections and other selected mixed infections, under strict medical supervision and antimicrobial‑stewardship principles. Composition…