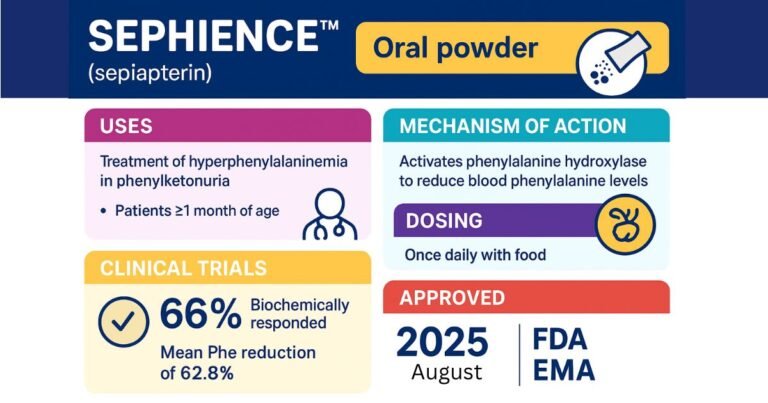
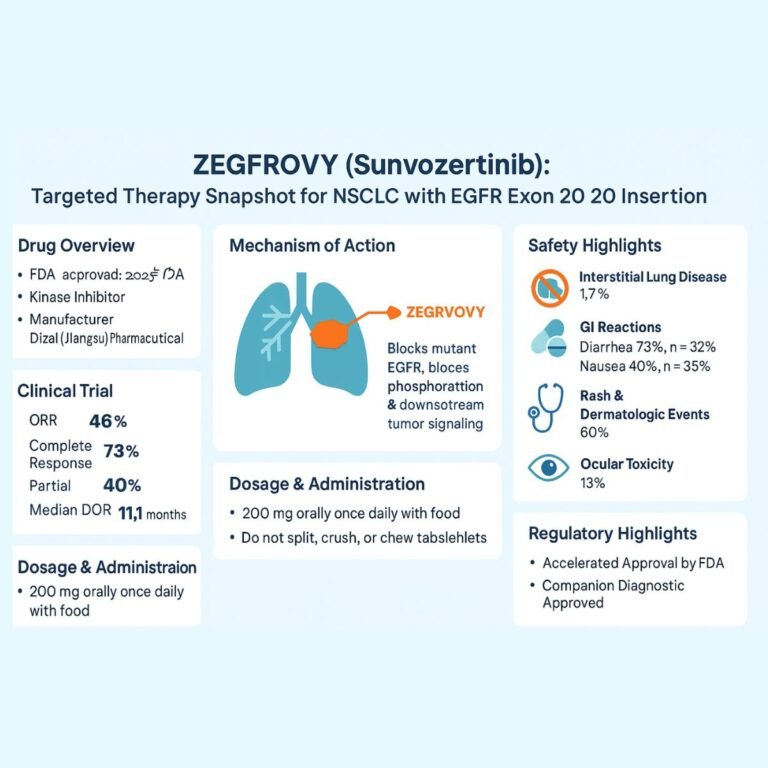
MODEYSO (dordaviprone) FDA Approval for Midline Glioma (DMG)

On August 6, 2025, the U.S. Food and Drug Administration (FDA) granted accelerated approval to MODEYSO (dordaviprone), a first-in-class protease activator for adult and pediatric patients aged one year and older with H3 K27M-mutant diffuse midline glioma (DMG) that has…