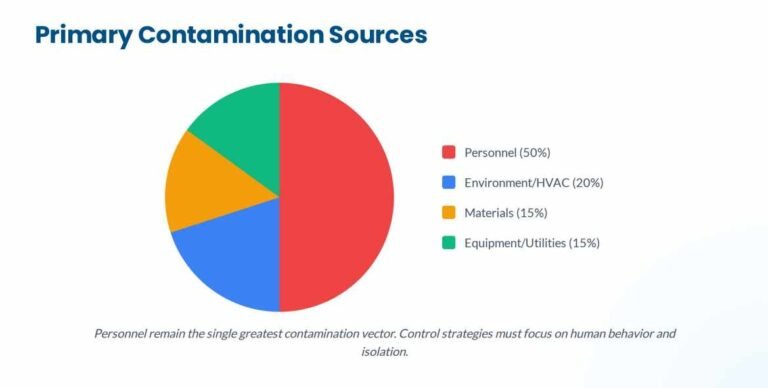
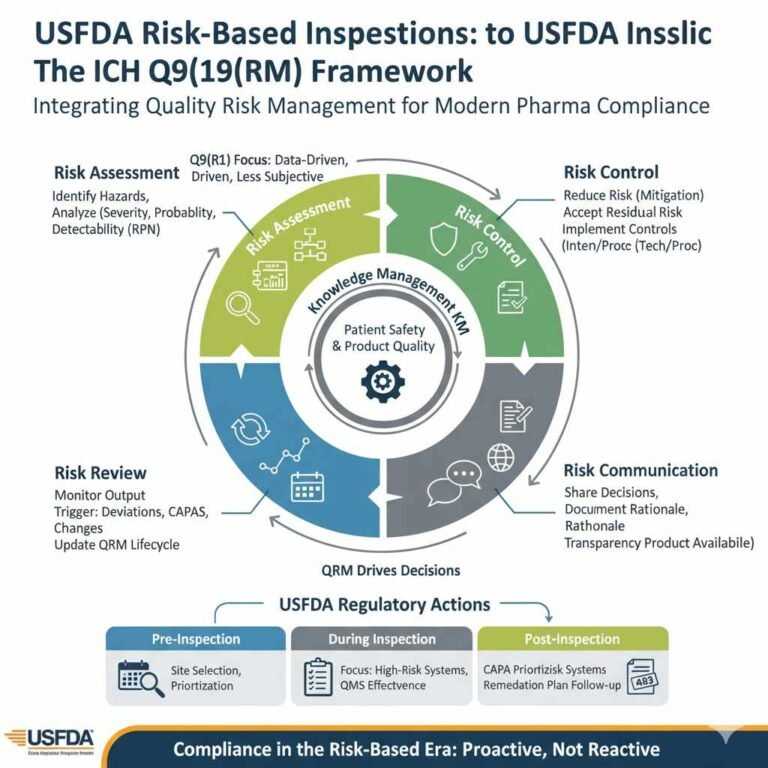
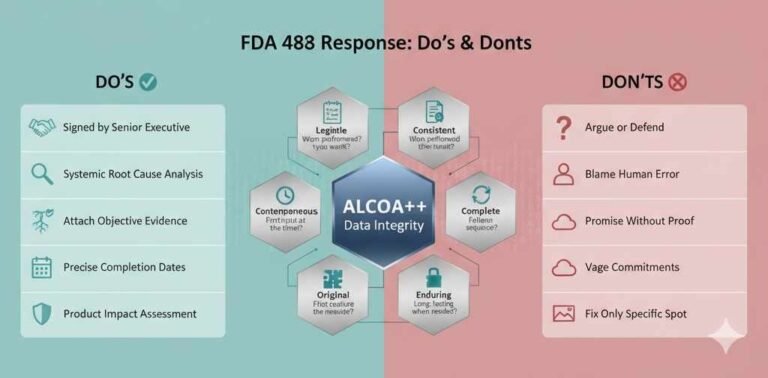
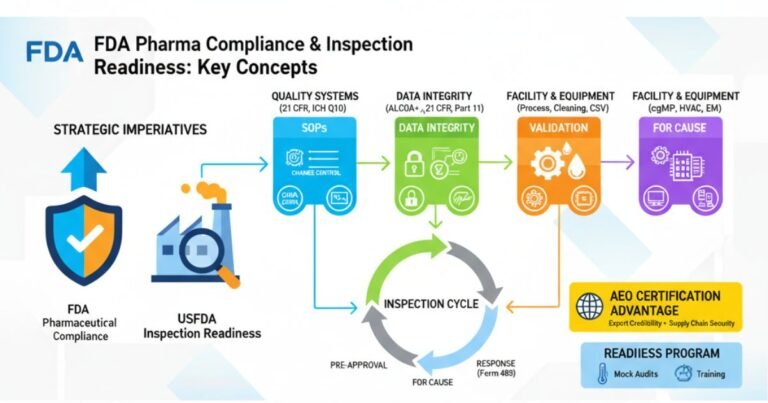
Rhapsido FDA Approval 2025: Comprehensive Analysis for Pharmaceutical Professionals
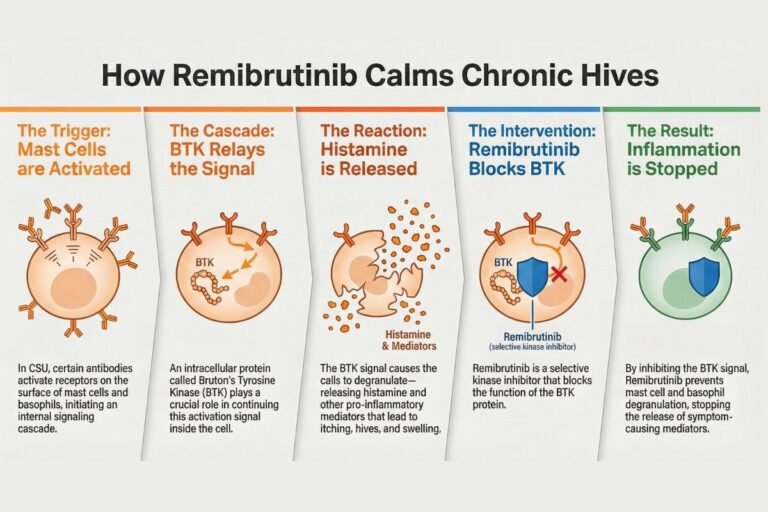
Rhapsido (remibrutinib) represents a significant advancement in chronic spontaneous urticaria (CSU) management following Rhapsido FDA approval on September 30, 2025. This first-in-class oral Bruton’s tyrosine kinase (BTK) inhibitor addresses a critical clinical gap for the estimated 1-2 million CSU patients failing…