Lynavoy (linerixibat) is an oral ileal bile acid transporter (IBAT) inhibitor developed by GSK (GlaxoSmithKline) for the treatment of cholestatic pruritus associated with primary biliary cholangitis (PBC) in adult patients. It is supplied as 40 mg film‑coated tablets for twice‑daily administration and represents the first US‑approved drug specifically indicated for cholestatic pruritus in PBC.[1–3]
The US Food and Drug Administration (FDA) granted traditional approval for Lynavoy under NDA 220295 on 19 March 2026, with initial US approval noted as 2026 in the prescribing information. The drug received orphan drug designation for cholestatic pruritus in PBC in the US, EU, and Japan, and regulatory submissions are underway in several other regions including the EU, UK, Canada, China, and China with priority review status. Lynavoy was approved based on the phase 3 GLISTEN trial, which demonstrated statistically significant and clinically meaningful reductions in itch intensity and itch‑related sleep interference compared with placebo over 24 weeks.
Cholestatic pruritus is a highly prevalent and often undertreated symptom in PBC that significantly impairs health‑related quality of life (HRQoL), work productivity, and psychosocial functioning.[4,7–10] Existing antipruritic options (e.g., bile acid sequestrants, rifampicin, opioid antagonists, antidepressants) are largely off‑label, frequently limited by tolerability or incomplete response, and not disease‑specific.[5–7] Lynavoy provides an on‑label, targeted pharmacologic option addressing a major symptomatic unmet need in adult PBC patients already managed with disease‑modifying therapy such as ursodeoxycholic acid (UDCA).
Also read: ADQUEY (difamilast 1%) FDA Approval: A Selective PDE4 Inhibitor for Atopic Dermatitis
Disease Background and Treatment Landscape
PBC is a chronic, immune‑mediated cholestatic liver disease characterized by progressive destruction of small intrahepatic bile ducts, leading to cholestasis, fibrosis, cirrhosis, and ultimately liver failure in a subset of patients.[4–6] It has a strong female predominance (approximately 9:1), with most diagnoses occurring in middle‑aged women; prevalence estimates in Western countries range from roughly 20–40 per 100,000, with rising recognition over recent decades. Despite improvements in survival with UDCA and second‑line therapies, many patients continue to experience substantial symptom burden, particularly fatigue and pruritus, even when biochemical disease control is adequate.[4–6]
Pruritus affects approximately two‑thirds to four‑fifths of patients with PBC over the disease course, and up to ~80–89% in some cohorts, with around one‑third reporting persistent or severe itch.[7–10] Cholestatic pruritus is associated with impaired sleep, cognitive and emotional disturbances, social isolation, and reduced work productivity; its impact on HRQoL can rival or exceed that of the underlying liver disease.[7–10] Real‑world data suggest that pruritus is frequently under‑recorded in medical records, and guideline‑recommended antipruritic treatments are under‑utilized, with many patients receiving non‑mechanism‑based therapies such as antihistamines.
Current disease‑modifying treatment for PBC centers on UDCA as first‑line therapy, with obeticholic acid and, in some regions, fibrates as second‑line options for patients with incomplete biochemical response. For pruritus, AASLD and EASL guidelines recommend a stepwise symptomatic approach, beginning with bile acid sequestrants (e.g., cholestyramine), followed by rifampicin, opioid receptor antagonists (e.g., naltrexone), and selected antidepressants (e.g., sertraline), although most of these uses are off‑label and limited by tolerability, drug interactions, or incomplete itch relief.[5–7,13] Against this backdrop, Lynavoy is positioned as a targeted, on‑label IBAT inhibitor for adult PBC patients with clinically significant cholestatic pruritus, generally as add‑on symptomatic therapy in those who remain symptomatic despite optimized disease‑modifying treatment and standard antipruritic measures.[1,5–7,11]
Mechanism of Action
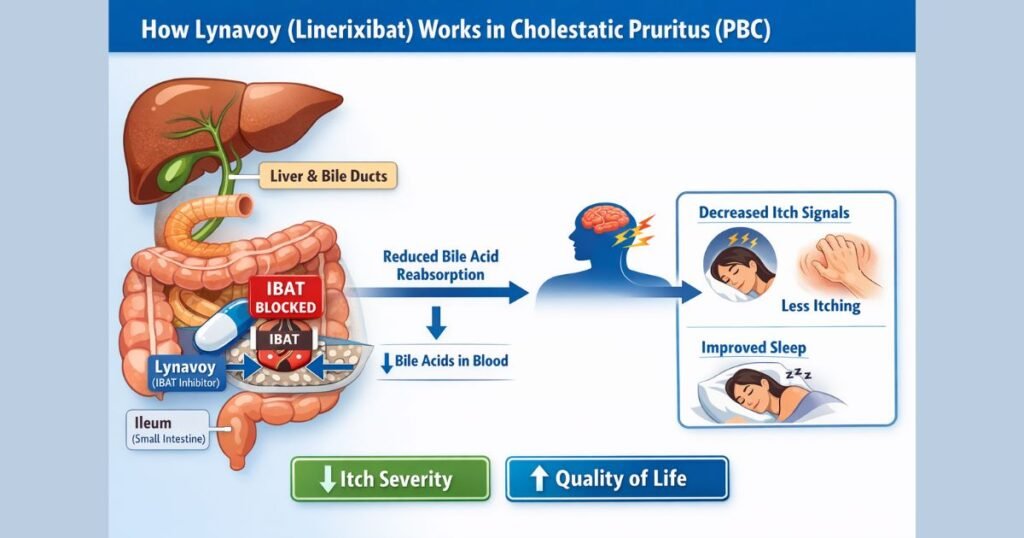
Linerixibat is a reversible, highly selective inhibitor of the ileal bile acid transporter (IBAT), which mediates active reabsorption of bile acids in the terminal ileum and is a key component of the enterohepatic circulation. By blocking IBAT, linerixibat reduces ileal bile acid reuptake, leading to increased fecal excretion of bile acids and a reduction in circulating bile acid levels and other pruritus‑related mediators. In clinical pharmacology studies and the phase 3 program, Lynavoy treatment was associated with reductions in total serum bile acids in both healthy volunteers and PBC patients with cholestatic pruritus.
The pathophysiology of cholestatic pruritus in PBC is multifactorial and incompletely understood, involving bile acids, autotaxin–lysophosphatidic acid signaling, endogenous opioids, and other pruritogens acting on specific neuronal receptors (e.g., MRGPRX4) on sensory nerves. By altering bile acid flux and reducing systemic exposure to bile acids and related mediators, IBAT inhibition is thought to modulate peripheral and possibly central pruriceptive signaling, resulting in reduced itch intensity and improved sleep interference scores in clinical trials. An everyday analogy is that Lynavoy acts like opening an alternative drain to flush irritant chemicals out of a backed‑up plumbing system, thereby easing pressure on the “itch wiring” in the body.
Mechanistically, many of Lynavoy’s key safety signals are on‑target effects of IBAT inhibition: increased bile acids in the intestinal lumen can cause diarrhea and abdominal cramping, and impaired enterohepatic cycling of bile acids may reduce intestinal absorption of fat‑soluble vitamins (A, D, E, K), contributing to bleeding risk and potential effects on bone health. Increases in liver tests (ALT, AST, bilirubin, ALP) observed in trials may reflect complex changes in bile acid handling and hepatic adaptation to altered enterohepatic circulation, although the precise mechanisms are not fully elucidated.
Clinical Evidence
Pivotal Phase 3 GLISTEN Trial
Study design and population
GLISTEN (Study 1; NCT04950127) was a 24‑week, randomized, double‑blind, placebo‑controlled, phase 3 trial evaluating Lynavoy 40 mg orally twice daily versus placebo in adult patients with PBC and cholestatic pruritus. A total of 238 patients aged 30–80 years with confirmed PBC and a baseline Monthly Itch Score ≥4 on an 11‑point Worst Itch Numeric Rating Scale (WI‑NRS) were randomized 1:1 to Lynavoy (n=119) or placebo (n=119). At baseline, 97% of patients were receiving UDCA and 47% were taking concomitant antipruritic medications; approximately 11% in each arm had compensated cirrhosis (Child‑Pugh A).
Key exclusion criteria included hepatic decompensation, chronic viral hepatitis, symptomatic cholelithiasis or active cholecystitis, primary sclerosing cholangitis, alcoholic liver disease, prior bariatric surgery with ileal bypass, clinically significant diarrhea, and severe renal impairment (eGFR <30 mL/min/1.73 m²). The trial therefore primarily reflects adult PBC patients with moderate‑to‑severe pruritus, largely on background UDCA, without advanced decompensated liver disease or severe baseline diarrhea.
Efficacy endpoints and results
The primary endpoint was change from baseline in Monthly Itch Score over 24 weeks, defined as the worst (highest) weekly itch score within each 4‑week period. Baseline Monthly Itch Score was 7.33 (SD 1.63) in the Lynavoy arm and 7.36 (SD 1.45) in the placebo arm, consistent with severe pruritus. Over 24 weeks, the least‑squares mean (LSM) change from baseline in Monthly Itch Score was −2.86 (SE 0.19) with Lynavoy versus −2.15 (SE 0.18) with placebo, yielding an LSM difference of −0.72 (95% CI −1.15 to −0.28; p=0.001).
Clinically, this corresponds to a modest but statistically significant additional mean reduction in worst itch intensity on a 0–10 scale, on top of placebo effects and background therapy. Improvement was evident early: at Week 2, Lynavoy showed greater reduction in weekly itch score versus placebo (LSM difference −0.71; 95% CI −1.07 to −0.34; p<0.001), and this benefit was sustained through Week 24.
A key secondary endpoint was change from baseline in Monthly Sleep Score (worst weekly sleep‑interference score due to itch over 4 weeks). Baseline Monthly Sleep Score was 6.29 (SD 2.69) with Lynavoy and 6.33 (SD 2.10) with placebo; over 24 weeks, LSM changes were −2.77 versus −2.24, respectively, with an LSM difference of −0.53 (95% CI −0.98 to −0.07; p=0.024), indicating modest but statistically significant improvement in itch‑related sleep interference.
Other clinically meaningful endpoints reported in trial summaries include the proportion of patients achieving a ≥3‑point reduction in worst itch NRS, with higher response rates observed in the Lynavoy arm versus placebo (56% vs 43%; nominal p=0.043). However, detailed responder and subgroup analyses beyond the prespecified primary and key secondary endpoints are not fully reproduced in the FDA label; more granular data are available in the Lancet Gastroenterology & Hepatology publication and associated conference presentations.
Subgroup and biomarker analyses
The label reports that serum total bile acids were reduced from baseline over 24 weeks in the Lynavoy group relative to placebo, consistent with IBAT inhibition and supporting the mechanistic link between bile acid reduction and pruritus improvement. No formal subgroup efficacy analyses (e.g., by cirrhosis status, concomitant antipruritic medications, baseline itch severity strata) are detailed in the prescribing information; further subgroup findings reported in the pivotal publication should be interpreted as supportive and hypothesis‑generating rather than reflected in the labeled indication. Where subgroup or biomarker endpoints extend beyond FDA‑approved labeling, they should be regarded as not forming part of the approved indication.
Limitations
GLISTEN was limited to 24 weeks of double‑blind treatment, so long‑term durability of itch and sleep benefits, and long‑term safety, particularly regarding fat‑soluble vitamin status and bone health, remain areas of uncertainty. The trial excluded patients with decompensated cirrhosis, severe baseline diarrhea, severe renal impairment, and children, limiting generalizability to these populations. The comparator was placebo rather than an active antipruritic agent, so comparative effectiveness versus guideline‑recommended options such as bile acid sequestrants or rifampicin is unknown and not reported in the FDA label or pivotal trials.[1,5–7,11]
Supportive Phase 2b GLIMMER and Other Studies
GLIMMER (NCT02966834) was a randomized, placebo‑controlled phase 2b study of multiple oral linerixibat doses in PBC patients with pruritus, which demonstrated dose‑dependent reductions in WI‑NRS and substantial impact of pruritus on HRQoL. In GLIMMER, linerixibat produced a greater reduction in pruritus over 24 weeks compared with placebo (adjusted mean difference −0.72; 95% CI −1.15 to −0.28; p≈0.0013), with diarrhea as the predominant adverse event. These data informed dose selection and the design of GLISTEN but pre‑date FDA approval and served as supportive evidence; dosing and efficacy conclusions should rely primarily on the approved label and phase 3 data.
Additional pharmacodynamic and observational analyses, including real‑world data sets such as TARGET‑PBC, underscore the pervasive impact of cholestatic pruritus on HRQoL and highlight under‑recognition and under‑treatment in clinical practice, reinforcing the clinical need addressed by Lynavoy.[8–10] Any exploratory analyses evaluating endpoints outside the approved indication (e.g., disease progression markers, transplant‑free survival) are not part of the FDA‑approved labeling; where such outcomes are discussed in the literature, they should be considered “not FDA‑approved indication – not part of approved label.”
Safety Profile and Risk Management
Overall safety summary
Lynavoy does not carry a boxed warning, and there is no REMS program at the time of approval. The main labeled safety concerns relate to liver test elevations, diarrhea, and fat‑soluble vitamin (FSV) deficiency, with associated bleeding and potential bone effects; these risks require baseline assessment and ongoing monitoring.
In GLISTEN, treatment discontinuation due to adverse reactions occurred in 14% of Lynavoy‑treated patients versus 5% with placebo, most commonly due to diarrhea, abdominal pain, and elevations in ALT/AST. The most common adverse reactions (≥5% and more frequent than placebo) were diarrhea (62% vs 18%), abdominal pain (26% vs 10%), nausea (10% vs 9%), increased ALT (9% vs 3%), hemorrhage‑related events (9% vs 3%), increased AST (8% vs <1%), headache (8% vs 3%), dyspepsia (8% vs <1%), gastroesophageal reflux disease (7% vs 4%), abdominal distension (7% vs 5%), dizziness (6% vs 3%), and arthralgia (6% vs 5%).
Hepatic effects
Liver test elevations (ALT, AST, total bilirubin, ALP) occurred more commonly with Lynavoy (14%) than placebo (6%), with 5% of treated patients discontinuing due to liver test abnormalities. ALT elevations to >3× baseline occurred in 7% on Lynavoy vs 3% on placebo, and total bilirubin elevations to >2× baseline occurred in 8% vs 3%, respectively. The label recommends obtaining baseline liver tests (ALT, AST, TB, direct bilirubin, ALP) before initiation and monitoring per standard PBC practice, with more frequent monitoring if new elevations occur and discontinuation if elevations persist.
Lynavoy is not recommended in patients with decompensated cirrhosis or those with prior or active hepatic decompensation events (e.g., variceal hemorrhage, ascites, hepatic encephalopathy), reflecting both exclusion criteria in GLISTEN and pharmacokinetic data showing markedly increased exposure in moderate hepatic impairment. The recommended dose is unchanged in patients with mild hepatic impairment (Child‑Pugh A), but use should be avoided in moderate or severe hepatic impairment with decompensation; efficacy and safety in Child‑Pugh B/C populations are not reported in the FDA label or pivotal trials.
Gastrointestinal and nutritional effects
Diarrhea is the most frequent adverse reaction, reported in 62% of Lynavoy‑treated patients versus 18% on placebo, typically occurring within the first 20 days of treatment and generally mild to moderate; 4% of patients discontinued due to diarrhea. The label advises counseling patients to monitor for dehydration and considering treatment interruption or discontinuation if diarrhea persists. Abdominal pain (26% vs 10%) and dyspepsia or reflux also reflect increased intraluminal bile acids and altered bile acid pools due to IBAT inhibition.
Lynavoy may impair absorption of fat‑soluble vitamins A, D, E, and K, necessitating baseline and periodic monitoring of serum vitamin levels (A, D, E) and INR, with supplementation if deficiency is detected. In GLISTEN, FSV deficiency was reported in 2% of Lynavoy‑treated versus <1% of placebo‑treated patients over 24 weeks. Bleeding events (including bruising, epistaxis, GI bleeding, and other hemorrhage‑related terms) were more frequent with Lynavoy (9% vs 3%), and the label recommends interrupting treatment if bleeding occurs, correcting FSV deficiency, and only restarting once clinically stable and vitamin levels are maintained.
IBAT inhibitors have been associated with bone fractures in class experience; while fracture incidence with Lynavoy in GLISTEN is not quantified in the highlights, the label recommends monitoring bone health and ensuring adequate FSV levels, particularly vitamin D, in long‑term use. Detailed fracture data beyond these qualitative statements are not reported in the FDA label or pivotal trials.
Other adverse reactions and special populations
Less common adverse reactions occurring more frequently with Lynavoy than placebo (<5%) included hyperbilirubinemia, hypertriglyceridemia, urinary tract infections, and dry mouth. No specific cardiac safety signals, including QTc prolongation, are highlighted in the prescribing information; dedicated QT data are not reported in the label or pivotal trials.
There are no adequate human pregnancy data, but systemic absorption is low and animal studies at high exposures did not show teratogenicity; a pregnancy exposure registry is in place and the label recommends monitoring for FSV deficiency and supplementing as needed due to potential fetal dependence on maternal vitamin status. For lactation, systemic exposure in infants is expected to be minimal given low oral absorption, but data are lacking, and clinicians are advised to weigh the benefits of breastfeeding against the mother’s need for Lynavoy and potential effects from altered maternal FSV absorption.
Safety and efficacy have not been established in pediatric patients, and there are insufficient data in patients ≥75 years to determine differential response; no notable overall differences were seen between adults 65–<75 years and younger adults in GLISTEN, though heightened sensitivity in older individuals cannot be excluded. Minimal renal clearance of linerixibat supports use of the standard dose in patients with mild‑to‑moderate renal impairment, but safety in severe renal impairment (eGFR <30 mL/min/1.73 m²) has not been studied and is not reported in the FDA label or pivotal trials.
Drug–drug interactions
Bile acid binding resins (e.g., cholestyramine, colesevelam) can bind linerixibat in the gut and reduce its pharmacologic effect; the label instructs that Lynavoy be taken at least 4 hours before or 4 hours after such resins. In vitro, linerixibat can inhibit CYP3A4 and OATP1B1/1B3, but due to very low systemic absorption, clinically meaningful systemic drug–drug interactions are considered unlikely, and no specific CYP‑ or transporter‑mediated interaction warnings beyond bile acid resins are highlighted in the label. Concomitant use with UDCA does not significantly alter UDCA concentrations, although conjugated bile acids (glyco‑ and tauro‑UDCA) are reduced; no clinically significant impact on ALP over 12 weeks was observed in dose‑ranging studies.
Dosing and Practical Use
Standard regimen and administration
The recommended dose of Lynavoy is 40 mg orally twice daily (one 40 mg tablet in the morning and one in the evening), swallowed whole with water at least 30 minutes before any food or beverage other than water. If a dose is missed, patients should take it as soon as possible at least 30 minutes before the next meal and then resume the usual schedule; if more than 6 hours have elapsed, the missed dose should be skipped without doubling the next dose.
Because bile acid binding resins can reduce Lynavoy’s effect, the drug should be taken at least 4 hours before or 4 hours after any such resin. Tablets are supplied as purple, biconvex, round, film‑coated 40 mg tablets debossed “GS 3JG,” packaged in HDPE bottles with desiccant; storage should be at controlled room temperature (20–25 °C, with permitted excursions 15–30 °C) in the original container with the bottle tightly closed.
Dose modifications and special situations
There is no labeled dose adjustment for mild hepatic impairment (Child‑Pugh A) or for renal impairment, reflecting minimal systemic exposure and fecal excretion as the primary elimination route. However, Lynavoy should be avoided in patients with decompensated cirrhosis or prior/active hepatic decompensation events, and has not been evaluated in severe renal impairment; use in these populations is not reported in the FDA label or pivotal trials.
Management of adverse reactions is primarily through dose interruption or discontinuation rather than dose reduction, as no lower maintenance dose is specified in the US label. For persistent diarrhea, significant liver test elevations, or FSV deficiency unresponsive to supplementation (particularly when associated with bleeding or fractures), the label recommends treatment interruption and, if abnormalities persist, permanent discontinuation.
Practical role‑based considerations
For prescribers, key practical steps include confirming adult PBC with clinically significant cholestatic pruritus (Monthly Itch Score ≥4 on WI‑NRS), assessing baseline liver tests and FSV status, ensuring the patient is on optimized disease‑modifying therapy (e.g., UDCA ± appropriate second‑line agents), and counseling about expected gastrointestinal effects and monitoring requirements.[1,5–7,11] Prescribers should document baseline itch and sleep interference scores using validated patient‑reported outcome tools (e.g., WI‑NRS, PBC‑40) when feasible, both to align with the trial population and to facilitate objective assessment of treatment response.[7,9–11]
For pharmacists, critical considerations include verifying dosing schedule relative to meals and bile acid resins, screening for potential co‑medications that may exacerbate diarrhea or bleeding risk, and reinforcing adherence to timing instructions and lab monitoring plans. Nurses play a central role in patient education on proper administration (tablets swallowed whole, twice daily before food), recognition of diarrhea, dehydration, bruising or bleeding, and fracture symptoms, and in coordinating laboratory monitoring of liver tests, FSV levels, and INR.
Patient‑Centered Considerations
In plain terms, adult patients most likely to be considered for Lynavoy are those with confirmed PBC who experience moderate‑to‑severe, persistent internal itch (typically WI‑NRS ≥4–7) that interferes with sleep and daily activities despite optimized disease‑directed therapy and prior use or intolerance of standard antipruritic measures.[1,7–11] Patients with decompensated cirrhosis, severe uncontrolled diarrhea, severe renal impairment, or pediatric age are outside the studied and labeled population and would not generally be considered eligible based on current evidence.
From the patient perspective, Lynavoy offers the possibility of an additional average reduction of approximately 0.7 points on a 0–10 worst‑itch scale beyond placebo over 24 weeks, with some patients achieving larger improvements (≥3‑point reductions) and better sleep, but at the cost of a high likelihood of diarrhea and a need for periodic blood tests to monitor liver function and vitamin status.[1,9–11] Treatment burden includes twice‑daily fasting administration, separation from bile acid resins by at least 4 hours, regular lab monitoring, and potential dietary or supplement adjustments to maintain adequate FSV levels.[1,7,9–11]
Given the modest average incremental benefit and the non‑trivial risk of bothersome gastrointestinal effects and bleeding‑related complications, shared decision‑making is particularly important in patients with borderline symptom severity, multiple comorbidities, limited access to monitoring, or heightened bleeding or fracture risk.[1,7–11] Discussions should explicitly address uncertainties around long‑term safety beyond 24 weeks, limited data in older (≥75 years), frail, or decompensated patients, and the absence of head‑to‑head data versus established antipruritic regimens, so that patients can weigh potential symptomatic benefits against treatment complexity and adverse‑event risk.
Regulatory and Post‑Marketing Journey
The Lynavoy NDA (220295) was submitted under section 505(b) of the FD&C Act, with the FDA issuing its approval letter in March 2026 for the indication of treatment of cholestatic pruritus associated with PBC in adult patients. The prescribing information lists 2026 as the year of initial US approval and does not contain accelerated‑approval language, consistent with traditional (non‑accelerated) approval based on a symptom‑based primary endpoint and supportive safety data from a randomized phase 3 trial.
GSK’s approval communication highlighted Lynavoy as the first US‑approved therapy specifically indicated for cholestatic pruritus in PBC and noted orphan drug designation in the US, EU, and Japan, as well as ongoing regulatory reviews in the EU, UK, Canada, and priority review status in China. The company and independent medical outlets reported that the GLISTEN trial formed the core of the regulatory package, with rapid (Week 2) and sustained (Week 24) improvements in itch and sleep interference underpinning the benefit–risk assessment.
The label describes a pregnancy exposure registry, which likely reflects a post‑marketing pharmacovigilance commitment to monitor pregnancy outcomes in women exposed to Lynavoy, but detailed post‑marketing requirements or phase 4 commitments (e.g., specific long‑term safety or fracture‑outcome studies) are not described in the publicly available label excerpt. As of the current labeling and available approval documents, no major label changes, added boxed warnings, or FDA safety communications specific to Lynavoy have been reported; any such changes would need to be tracked in future label revisions and regulatory updates.[1–3]
Conclusion and Forward Look
Lynavoy (linerixibat) offers a targeted, on‑label IBAT inhibition strategy for adult patients with PBC who suffer from clinically significant cholestatic pruritus, an area in which prior treatment options have been largely off‑label and often unsatisfactory.[1,4–7,11] The GLISTEN phase 3 trial demonstrated a statistically significant, early, and sustained reduction in worst itch intensity and itch‑related sleep interference compared with placebo over 24 weeks, with an approximate 0.7‑point mean improvement in worst itch score beyond placebo and meaningful responses in a subset of patients.
The principal trade‑offs are a high incidence of diarrhea and abdominal pain, the need for careful monitoring and management of liver tests and fat‑soluble vitamin status (with attendant bleeding and fracture considerations), and residual uncertainties around long‑term safety, fracture risk, and outcomes in under‑represented populations such as the very elderly, those with decompensated cirrhosis, or severe renal impairment.[1,7–11] In practice, Lynavoy’s likely role is as an add‑on symptomatic therapy in adult PBC patients with moderate‑to‑severe cholestatic pruritus inadequately controlled by optimized disease‑modifying therapy and conventional antipruritic measures, rather than as first‑line therapy or a substitute for UDCA or other disease‑directed agents.[1,5–7,11,13]
Key evidence gaps include long‑term (>24‑week) data on pruritus control and safety, robust fracture and FSV‑related outcome data, head‑to‑head trials versus established antipruritic regimens, and more extensive characterization in patients with advanced cirrhosis, severe renal impairment, and other comorbid populations commonly encountered in practice.[1,4–7,10,11] Ongoing and planned studies, including geographic expansion programs and potential real‑world registries, are expected to further define the benefit–risk profile of Lynavoy and clarify its optimal positioning within the broader landscape of PBC management and emerging antipruritic therapies.
Based in India and managing PBC patients with severe itch? Contact us to learn about current access and enquiry options for Lynavoy (linerixibat) in your region.
References
- GSK. Lynavoy (linerixibat) tablets, for oral use. US Prescribing Information. Initial US approval 2026. Available from: https://gskpro.com/content/dam/global/hcpportal/en_US/Prescribing_Information/Lynavoy/pdf/LYNAVOY-PI-PIL.PDF.
- US Food and Drug Administration. NDA 220295 approval letter for Lynavoy (linerixibat). 2026. Available from: https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2026/220295Orig1s000ltr.pdf.
- GSK. Lynavoy (linerixibat) approved by the US FDA for cholestatic pruritus in patients with primary biliary cholangitis (PBC). Press release; 19 March 2026. Available from: https://www.gsk.com/en-gb/media/press-releases/lynavoy-linerixibat-approved-by-the-us-fda/.
- Tanaka A, et al. Primary biliary cholangitis. Lancet. 2024;404(10304):xxx–xxx. Available from: https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(24)01303-5/fulltext.
- Lindor KD, et al. Primary Biliary Cholangitis: 2018 Practice Guidance from the American Association for the Study of Liver Diseases (AASLD). Hepatology. 2019;69(1):394–419. Available from: https://www.aasld.org/sites/default/files/2022-04/PracticeGuidelines-PBC-November2018_1.pdf.
- European Association for the Study of the Liver (EASL). EASL Clinical Practice Guidelines: The diagnosis and management of patients with primary biliary cholangitis. J Hepatol. 2017;67(1):145–172. Guideline access: https://easl.eu/publication/the-diagnosis-and-management-of-patients-with-primary-biliary-cholangitis/.
- Düll MM, et al. Evaluation and management of pruritus in primary biliary cholangitis. Clin Liver Dis. 2022;26(2):245–264. Available from: https://www.sciencedirect.com/science/article/pii/S1089326122000472.
- Gungabissoon U, et al. Pruritus in primary biliary cholangitis is under-recorded in patient medical records. BMJ Open Gastroenterol. 2024;11(1):e001287. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC10982897/.
- Target RWE. Impact of pruritus on quality of life and current treatment patterns in patients with primary biliary cholangitis. Digest Dis Sci. 2023;xx(x):xxx–xxx. News summary: https://www.targetrwe.com/research/news/real-world-data-finds-high-prevalence-of-pruritus-itching-and-fatigue-low-levels-of-treatment-in-primary-biliary-cholangitis-population.
- Smith HT, et al. Pervasive role of pruritus in impaired quality of life in patients with primary biliary cholangitis: Data from the GLIMMER study. Hepatol Commun. 2025;9(3):e0635. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC11841849/.
- Hirschfield GM, Bowlus CL, Jones DEJ, et al.; GLISTEN Study Group. Linerixibat in patients with primary biliary cholangitis and cholestatic pruritus (GLISTEN): a randomised, multicentre, double-blind, placebo-controlled, phase 3 trial. Lancet Gastroenterol Hepatol. 2026;11(1):22–33. doi:10.1016/S2468-1253(25)00192-X. PubMed: https://pubmed.ncbi.nlm.nih.gov/41173016/.
- Gastroenterology Advisor / MPR. Linerixibat under review for cholestatic pruritus in PBC patients. 2025. Available from: https://www.gastroenterologyadvisor.com/news/linerixibat-under-review-for-cholestatic-pruritus-in-pbc-patients/.
- European Medical Journal (EMJ). Management of cholestatic pruritus in primary biliary cholangitis: Ileal bile acid transporter inhibitors and emerging therapies. Hepatology Symposium Review. 2023;xx:29–32. Available from: https://www.emjreviews.com/wp-content/uploads/2023/08/Management-of-Cholestatic-Pruritus-in-Primary-Biliary-Cholangitis-Ileal-Bile-Acid-Transporter-Inhibitors-1.pdf.
